 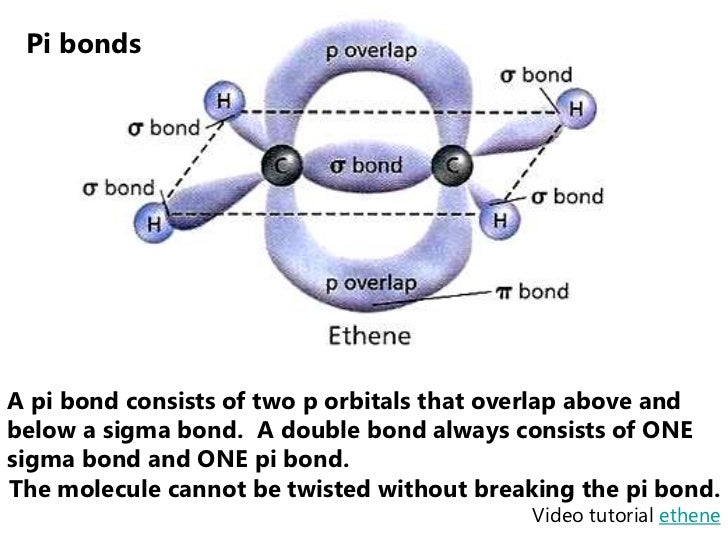
It is formed by the parallel or lateral overlapping of the atomic orbitals. A pi bond is a type of covalent bond that exists between atoms where the electrons are on top and bottom of the axis connecting the nuclei of the joined atoms. It gets a bit more complex when you get to University level chemistry since a new theory steps in, but I won’t give away the game yet (there’s something really fun about chem during which your previous concepts are shattered and rebuilt).Įdit – image here, although I think the hands explanation might work without it. Sigma bond is a chemical bond formed by the linear or co-axial overlapping of the atomic orbitals of two atoms. 
If your lateral overlap is disrupted, i.e the hands don’t cover one another you tend to lose pi bonding – this tends to happen if the system is non-planar, which can sometimes be caused by bulky groups disrupting the carbon skeleton. 
So in terms of electron density around the double or triple bond you can expect to see two rings (one above and one below the sigma bond) for alkenes, whereas for alkynes it’s a cylinder. Pi bonds are usually formed using the p orbitals in Carbon – so alkenes have one pi bond (since two p orbitals are used in sp2 hybridization), whereas alkynes have two pi bonds (only one p orbital used in sp hybridization). If you were to look at one hand straight on you shouldn’t be able to see the other hand. They can be a distance apart, but they have to be facing each other, and in the same plane. Pi orbitals on the other hand are like having two hands waving at one another. Overarching idea is that your sigma bonds are caused by linear overlap of orbitals – kind of like a handshake – the hands grasp each other, whilst the orbitals overlap really nice and tight next to one another too. I’ve used carbon as a model system- but you can get sigma and pi bonds in other compounds as well. Ahoy – Ruhina has done this pretty well, so thought best to add a drawing – hopefully it is clear enough. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
